General Overview
The Molecular Cell Biology Research Group has a special focus on membrane transporter proteins, pluripotent stem cells, and stem cell differentiation, with the aim of exploring their potential role in the pathophysiology of various diseases, including common disorders, such as type II diabetes and gout, as well as less frequent diseases, such as schizophrenia and DiGeorge syndrome. Members of the Group employ a wide range of molecular biological, biochemical, and cell biology approaches to establish various cellular models for drug screening, diagnostic purposes, and disease modelling. The ultimate goal of the pursued research is to reveal novel therapeutic targets and to develop novel diagnostic methods for the studied disorders.
The interdisciplinary nature of the topic encourages the Research Group to collaborate with a wide range of partners. Fruitful collaborations marked with joint publications have been established with research groups within the HUN-REN network (e.g., Institute of Organic Chemistry, Institute of Materials and Environmental Chemistry, Institute of Experimental Medicine), research groups at universities (e.g., Dept. of Psychiatry and Psychotherapy, Dept. of Biophysics and Radiation Biology, Dept. of Physiology, Heart and Vascular Centre at the Semmelweis University; Dept. of Biochemistry and Molecular Biology, Dept. of Pharmacology and Pharmacotherapy at the University of Szeged; Department of Biophysics and Cell Biology at the University of Debrecen; Department of Biochemistry at the Eötvös Loránd University), international collaborators (e.g., NIH, Bethesda, USA; Lunenfeld-Tanenbaum Research Institute, Mount Sinai Hospital, Toronto; Laboratoire d’Etude de l’Unité Neurovasculaire et Innovation Thérapeutique, Paris; Institute of Human Genetics, Universität Heidelberg; School of Biomedical Sciences at the Hong Kong University), and industrial partners (e.g., Solvo Biotechnology; Aktogen Hungary, Ltd; Cellpharma, Ltd; Pluristyx, Canada).
The research group consists of three closely collaborating subunits: the Laboratory of Cell Polarity and Trafficking, the Human Pluripotent Stem Cell Lab, and the Flow Cytometry Core Facility.
Laboratory of Cell Polarity and Trafficking
Head of the laboratory: László Homolya, D.Sc. (MTMT ID: 10010446)
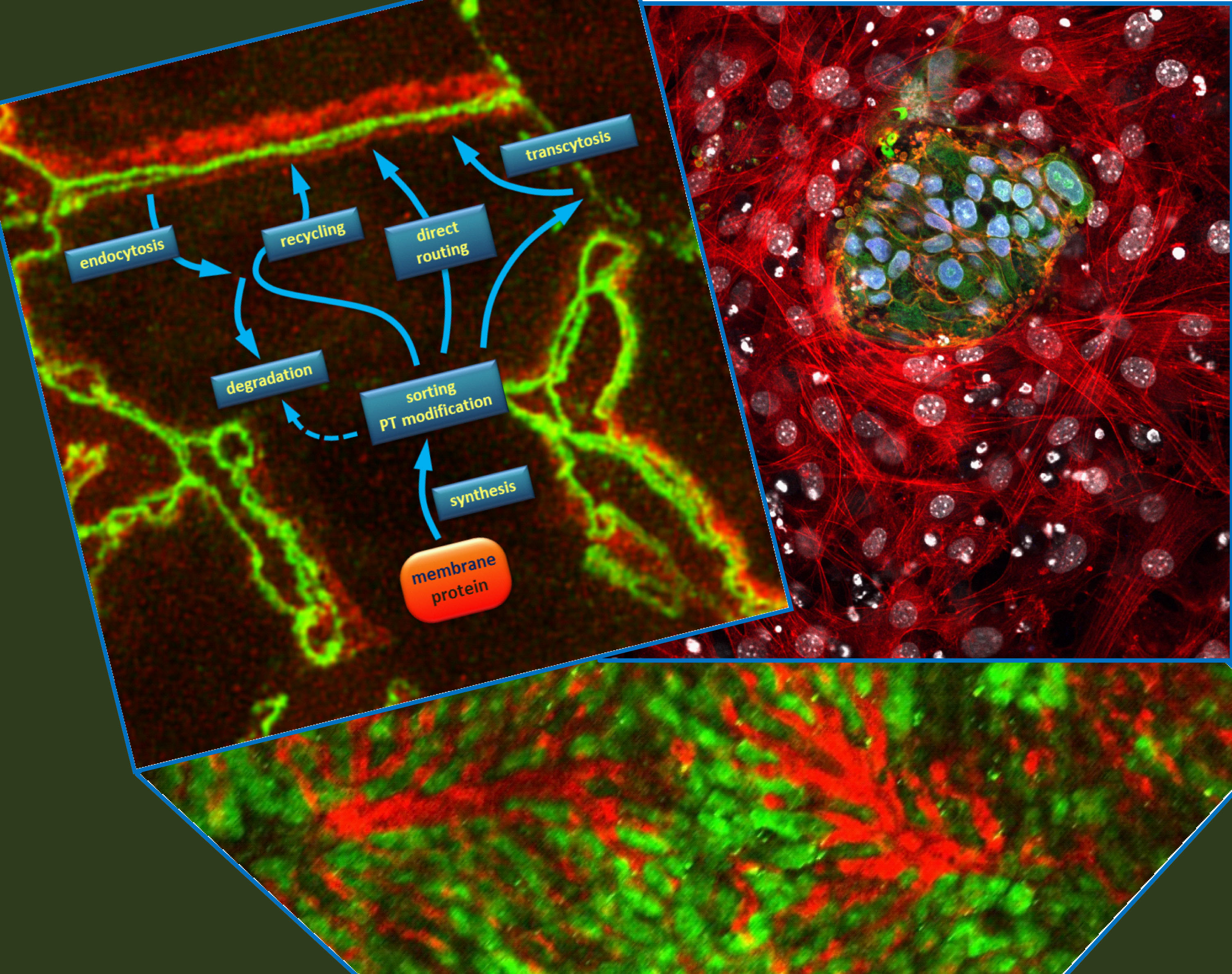
The physiological barriers of our body are formed by polarized epithelial and endothelial cells. Membrane transporter proteins, localized in the membrane of these cells in a regulated fashion, control the translocation of various substances, such as nutrients, toxins, and drug molecules. The research of the Lab focuses on the better understanding of the establishment and maintenance of cell polarity; the targeting and trafficking of transporter proteins to the proper membrane compartment; and how the genetic alterations – especially disease-causing mutations – in the transporters affect their cellular routing and consequently their function. In addition, the mechanisms regulating the formation of special forms of cell polarity, such as hepatocyte and neural cell polarity, are studied in detail.
Human Pluripotent Stem Cell Lab
Head of the laboratory: Ágota Apáti, D.Sc. (MTMT ID: 10027529)
Pluripotent stem cells can in vitro be differentiated into any cell type of the body. The Human Pluripotent Stem Cell Lab gains knowledge on human stem cell generation and differentiation especially in the direction of cardiac, neuronal, endothelial, mesenchymal, and hepatic lineages. The Lab has strong experience in studying membrane transporters and calcium signaling in human pluripotent stem cells and their differentiated progenies. The recent focus is on developing induced pluripotent stem cell-based disease models for More recently, induced pluripotent stem cell-based disease models and transgenic cell reporter systems are being developed to study the cellular pathophysiology of human disorders such as schizophrenia, DiGeorge syndrome, arteriosclerosis, and type II diabetes.
Flow Cytometry Core Facility
Head of the laboratory: György Várady, Ph.D. (MTMT ID: 10015754)
The Flow Cytometry lab serves as a core facility and research laboratory. Our cell sorter allows us to generate single cell clones and homogeneous cell lines based on the cellular fluorescence intensities. In addition, cytometers are used for the assessment of protein levels, transfection efficiency, and multidrug resistance, as well as for cell cycle analysis. Our main research area includes the functional investigation of ABC transporters using fluorescent dyes and the characterization of multifactorial diseases (gout, type 2 diabetes) by the analysis of the membrane proteins in red blood cells.
Main instruments:
FACSAria III cell sorter with Automated Cell Deposition Unit (ACDU);
Attune Nxt with 3 (viola, blue, red) and 4 (viola, blue, yellow, red) lasers with autosampler;
FACSCanto II with 3 (viola, blue, red) lasers with High Throughput Sampler (HTS)
Selected recent publications:
- Mózner O, Zámbó B, Bartos Z, Gergely A, Szabó KS, Jezsó B, Telbisz Á, Várady G, Homolya L, Hegedűs T, Sarkadi B. Expression, Function and Trafficking of the Human ABCG2 Multidrug Transporter Containing Mutations in an Unstructured Cytoplasmic Loop. Membranes 13(10):822. doi: 10.3390/membranes13100822 (2023)
- Bartos Z. and Homolya L.: Identification of specific trafficking defects of naturally occurring variants of the human ABCG2 transporter. Front Cell Dev Biol 9:615729. doi: 10.3389/fcell.2021.615729 (2021)
- Sarkadi B, Homolya L, Hegedűs T. The ABCG2/BCRP transporter and its variants – from structure to pathology. FEBS Lett. 594(23):4012-4034. doi: 10.1002/1873-3468.13947 (2020)
- Lilienberg J, Apáti Á, Réthelyi JM, Homolya L. Microglia modulate proliferation, neurite generation and differentiation of human neural progenitor cells. Front Cell Dev Biol. 10:997028. doi: 10.3389/fcell.2022.997028. (2022)
- Lilienberg J, Hegyi Z, Szabó E, Hathy E, Málnási-Csizmadia A, Réthelyi JM, Apáti Á, and Homolya L. Pharmacological Modulation of Neurite Outgrowth in Human Neural Progenitor Cells by Inhibiting Non-muscle Myosin II. Front Cell Dev Biol. 9:719636 doi: 10.3389/fcell.2021.719636. eCollection (2021)
- László L, Maczelka H, Takács T, Kurilla A, Tilajka Á, Buday L, Vas V, Apáti Á. A Novel Cell-Based Model for a Rare Disease: The Tks4-KO Human Embryonic Stem Cell Line as a Frank-Ter Haar Syndrome Model System. Int J Mol Sci. 2022 Aug 8;23(15):8803. doi: 10.3390/ijms23158803 (2022)
- Reé D, Fóthi Á, Varga N, Kolacsek O, Orbán TI, Apáti Á. Partial Disturbance of Microprocessor Function in Human Stem Cells Carrying a Heterozygous Mutation in the DGCR8 Gene. Genes 13(11):1925. doi: 10.3390/genes13111925. (2022)
- Hathy E, Szabó E, Varga N, Erdei Z, Tordai C, Czehlár B, Baradits M, Jezsó B, Koller J, Nagy L, Molnár MJ, Homolya L, Nemoda Z, Apáti Á, Réthelyi JM. Investigation of de novo mutations in a schizophrenia case-parent trio by induced pluripotent stem cell-based in vitro disease modeling: convergence of schizophrenia- and autism-related cellular phenotypes. Stem Cell Res Ther. 11(1):504. doi: 10.1186/s13287-020-01980-5 (2020)
- Apáti Á, Varga N, Berecz T, Erdei Z, Homolya L, Sarkadi B. Application of human pluripotent stem cells and pluripotent stem cell-derived cellular models for assessing drug toxicity. Expert Opin Drug Metab Toxicol. 15(1):61-75. doi: 10.1080/17425255.2019.1558207 (2019)
- Kulin A, Kucsma N, Bohár B, Literáti-Nagy B, Korányi L, Cserepes J, Somogyi A, Sarkadi B, Szabó E, Várady G. Genetic Modulation of the GLUT1 Transporter Expression-Potential Relevance in Complex Diseases. Biology 11(11):1669. doi: 10.3390/biology11111669 (2022). PMID: 36421383; PMCID: PMC9687623.
- Szabó E, Kulin A, Jezsó B, Kucsma N, Sarkadi B, Várady G. Selective Fluorescent Probes for High-Throughput Functional Diagnostics of the Human Multidrug Transporter P-Glycoprotein (ABCB1). Int J Mol Sci. 23(18):10599. doi: 10.3390/ijms231810599 (2022)
- Szabó E, Kulin A, Mózner O, Korányi L, Literáti-Nagy B, Vitai M, Cserepes J, Sarkadi B, Várady G. Potential role of the ABCG2-Q141K polymorphism in type 2 diabetes. PLoS One. 16(12):e0260957. doi: 10.1371/journal.pone.0260957 (2021)
Highly cited publications from previous years
- Homolya L, Fu D, Sengupta P, Jarnik M, Gillet JP, Vitale-Cross L, Gutkind JS, Lippincott-Schwartz J, and Arias IM: LKB1/AMPK and PKA Control ABCB11 Trafficking and Polarization in Hepatocytes.PLOS ONE 9:(3) p. e91921. (2014)
- Homolya L, Orbán TI, Csanády L, and Sarkadi B. Mitoxantrone is expelled by the ABCG2 multidrug transporter directly from the plasma membrane. Biochim Biophys Act. 1808(1): 154-63 (2011)
- Apáti A, Orbán TI, Varga N, Németh A, Schamberger A, Krizsik V, Erdélyi-Belle B, Homolya L, Várady G, Padányi R, Karászi E, Kemna EW, Német K, Sarkadi B. High level functional expression of the ABCG2 multidrug transporter in undifferentiated human embryonic stem cells. Biochim Biophys Acta. 1778(12):2700-9. doi: 10.1016/j.bbamem.2008.08.010 (2008)
- Sarkadi B, Homolya L, Szakács G, and Váradi A. Human multidrug resistance ABCB and ABCG transporters: participation in a chemoimmunity defense system. Physiol Reviews 86: 1179-1236, (2006)
- Homolya L, Steinberg TH, and Boucher RC. Cell to cell communication in response to mechanical stress via bilateral release of ATP and UTP in polarized epithelia. Cell Biol., 150: 1349-1359, (2000)
- Homolya L, Watt CW, Lazarowski ER, Koller BH, and Boucher RC. Nucleotide-regulated calcium signaling in lung fibroblasts and epithelial cells from normal and P2Y2 receptor (-/-) mice. Biol. Chem., 274: 26454-26460 (1999)
- Lazarowski, E. R., Homolya, L., Boucher, R. C., and Harden, T. K.: Direct demonstration of mechanically induced release of UTP and its implication for uridine nucleotide receptor activation. Biol. Chem., 272: 24348-24354 (1997)
- Homolya L, Holló Zs, Germann U, Pastan I, Gottesman MM, and Sarkadi B. Fluorescent cellular indicators are extruded by the multidrug resistance protein. Biol. Chem., 268: 21493-21496 (1993)
Leader
László Homolya