General research interest
Our research activity is focused on the general aspects of protein structure, structural dynamics and conformational flexibility. We are particularly interested in the role of protein-protein interactions and allosteric conformational transitions in different biological processes. Our model proteins are mainly allosteric enzymes that can self organize to form sophisticated multi-molecular structures. In our research we apply both in silico modeling and wet laboratory experiments in a concerted manner.
Major research projects
Activation and regulation of the complement system
The complement system (CS) is a major effector arm of the innate immune system. The CS consists of about 40 protein molecules, and it is capable of recognizing, labeling and eliminating dangerous structures such as pathogen microorganisms. The major components of the CS are serine protease enzymes that activate each-other in a cascade-like manner. An intact complement system is essential for maintaining the immune homeostasis of the body; however, its uncontrolled, pathological activation may result in self-tissue damage. Recently it has been turned out that the pathological complement activation contributes to the development of numerous disease conditions. Our research concentrates on the study of the early serine proteases that initiate the complement activation. We recombinantly express the proteases in different expression systems and characterize their structural, biochemical and enzymological properties. Special emphasis is laid on the study of the natural and artificial inhibitors of the complement serine proteases. We determined the 3D structure of several complement serine proteases and characterized their enzymatic properties (Fig. 1). We clarified the exact mechanism of the lectin pathway activation, disproving a long-held previous model. Recently, we have made fundamental discoveries about the activation of the alternative pathway and about the cross-talk between the lectin and the alternative pathways (Fig. 2).
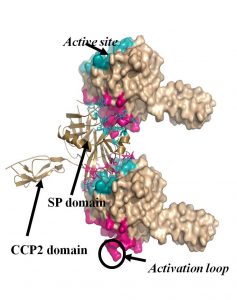
Fig. 1: The 3D structure of autoactivating MASP-2 molecules
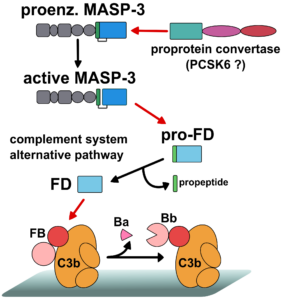
Fig. 2
In collaboration with the Department of Biochemistry of the Eötvös University we developed lectin pathway specific inhibitors. These inhibitors have proven to be very useful basic research tools and they can also serve as the basis for drug development. We founded a spin-off company (EvolVeritas Ltd.) to be able to involve venture capital in the drug development process.
The effect of mutations and posttranslational modifications on the structure and dynamics of proteins
Human genome studies have revealed an immense number of non-synonymous single-nucleotide variations, indicating the presence of large numbers of protein mutations. Many of these mutations are associated with disease. In the future, personalized medicine will involve the sequencing of individual genomes so that treatment options can be given based on the analysis of the genome. Thus, methods allowing us to interpret individual variations are in demand. Mapping mutations to 3D structures of proteins can give us important insight into the functioning of the protein, and how a mutation affects it. Posttranslational modifications (like phosphorylation) have a similar effect; these are essential for signaling, but the function of many is still unknown. Mutations and posttranslational modifications affect the function of proteins in several ways, including modulating the conformational dynamics of the protein. We reveal these effects on a number of proteins, including enzymes and regulatory proteins, using simulations that can describe the functional dynamics of proteins, including structural changes and changes in dynamical processes.
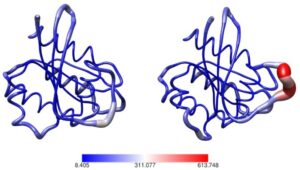
Fig. 3
Identification and characterization of interactions between proteins participating in signal transduction processes leading to cancer and neurological disorders
The essential physiological processes of the living cells are based on the sophisticated network of biological macromolecules, mainly that of proteins. The signal transduction pathways are mediated by different protein-protein interactions (e.g. strong or weak; direct or indirect), that regulate the functions of the allosteric enzymes.
The allosteric regulation, based on the conformational flexibility of proteins, is of great significance in the function of enzymes; and we have previously gained insight into such regulatory mechanisms by using isolated proteins. Based on our knowledge, in the frame of the present project, we would like to identify and characterize enzyme-enzyme interactions at the molecular level that are involved in signal transduction processes having pathophysiological relevance.
In the focus of the project are protein kinases (ROCK1, ROCK2, LIMK1 and LIMK2) which have multiple roles in cellular processes. Our main aim is to find specific protein-protein interactions which can be individually inhibited by allosteric modulators, therefore very specific drug molecules can be designed.
We have previously determined the 3D structure of ROCK2 with and without bound RhoA using Small-Angle X-ray Scattering (Fig. 4) and established a new activation model.
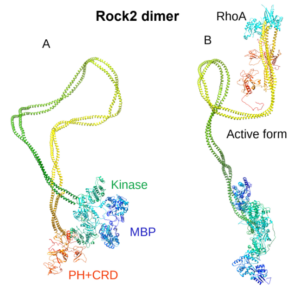
Fig. 4
We have also shown that an allosteric binding site is involved in the interaction between ROCK2 and Amyloid Precursor Protein (APP) leading to the unwanted phosphorylation of APP which leads to increases Aβ production, thus amyloid plaque leading to Alzheimer’s disease. Our results opened the way to attack the allosteric site to prevent APP phosphorylation at Thr654 by ROCK2 without inhibiting the activity of ROCK2 towards its other substrates.
Group members
- Attila Baksa, junior research fellow
- Júlia Balczer, technician
- József Dobó, PhD, senior research fellow
- Bence Farkas, PhD student
- Péter Gál, PhD, DSc, group leader, scientific advisor
- István Hajdú, PhD, research fellow
- Róbert Martinusz, graduate student
- Olivér Nagy, graduate student
- Judit Pataki, technician
- Andrea Párisné Kocsis, PhD, research fellow
- Erika Szabadi, laboratory assistant
- Barbara Végh, PhD, research fellow
- Péter Závodszky, member of the Hungarian Academy of Sciences, professor emeritus
Phd students
- László Beinrohr (2010)
- Gergely Gyimesi (2011)
- Balázs Major (2012)
- Andrea Párisné Kocsis (2012)
- Márton Megyeri (2012)
- Katalin Paréj (2015)
- Dániel Györffy (2015)
- Ráchel Sajó (2016)
- Gábor Oroszlán (2018)
- Ráhel Dani (2022)
Selected publications
- Németh Z, Debreczeni ML, Kajdácsi E, Dobó J, Gál P, Cervenak L. (2023) Cooperation of Complement MASP-1 with Other Proinflammatory Factors to Enhance the Activation of Endothelial Cells. Int J Mol Sci. 24(11):9181.
- Dani R, Oroszlán G, Martinusz R, Farkas B, Dobos B, Vadas E, Závodszky P, Gál P, Dobó J. (2023) Quantification of the zymogenicity and the substrate-induced activity enhancement of complement factor D. Front Immunol. 14:1197023.
- Golomingi M, Kohler J, Jenny L, Hardy ET, Dobó J, Gál P, Pál G, Kiss B, Lam WA, Schroeder V. (2022) Complement lectin pathway components MBL and MASP-1 promote haemostasis upon vessel injury in a microvascular bleeding model. Front Immunol. 13:948190.
- Nagy ZA, Héja D, Bencze D, Kiss B, Boros E, Szakács D, Fodor K, Wilmanns M, Kocsis A, Dobó J, Gál P, Harmat V, Pál G. (2022) Synergy of protease binding sites within the ecotin homodimer is crucial for inhibition of MASP enzymes and for blocking lectin pathway activation. J Biol Chem. 25:101985.
- Oroszlán G, Dani R, Végh BM, Varga D, Ács AV, Pál G, Závodszky P, Farkas H, Gál P, Dobó J. (2021) Proprotein Convertase Is the Highest-Level Activator of the Alternative Complement Pathway in the Blood. J Immunol. 206, 2198-2205.
- Hajdú I, Szilágyi A, Végh BM, Wacha A, Györffy D, Gráczer É, Somogyi M, Gál P, Závodszky P. (2020) Ligand-induced conformational rearrangements regulate the switch between membrane-proximal and distal functions of Rho kinase 2. Commun Biol. 3(1):721.
- Nagy ZA, Szakács D, Boros E, Héja D, Vígh E, Sándor N, Józsi M, Oroszlán G, Dobó J, Gál P, Pál G. (2019) Ecotin, a microbial inhibitor of serine proteases, blocks multiple complement dependent and independent microbicidal activities of human serum. PLoS Pathog. 15:e1008232.
- Szakács D, Kocsis A, Szász R, Gál P, Pál G. (2019) Novel MASP-2 inhibitors developed via directed evolution of human TFPI1 are potent lectin pathway inhibitors. J Biol Chem. 294, 8227-8237.
- Paréj K, Kocsis A, Enyingi C, Dani R, Oroszlán G, Beinrohr L, Dobó J, Závodszky P, Pál G, Gál P. (2018) Cutting Edge: A New Player in the Alternative Complement Pathway, MASP-1 Is Essential for LPS-Induced, but Not for Zymosan-Induced, Alternative Pathway Activation. J Immunol. 200, 2247-2252.
- Dobó J, Szakács D, Oroszlán G, Kortvely E, Kiss B, Boros E, Szász R, Závodszky P, Gál P, Pál G. (2016) MASP-3 is the exclusive pro-factor D activator in resting blood: the lectin and the alternative complement pathways are fundamentally linked. Sci Rep. 6:31877.
- Kidmose, R.T., Laursen, N.S., Dobó, J., Kjaer, T.R., Sirotkina, S., Yatime, L., Sottrup-Jensen, L., Thiel, S., Gál, P. and Andersen, G.R. (2012) ¬Structural basis for activation of the complement system by C4 cleavage. Proc. Natl. Acad. Sci. USA, 109, 15425-15430
- Héja, D., Kocsis, A., Dobó, J., Szilágyi, K., Szász, R., Závodszky, P., Pál, G. and Gál, P. (2012) Revised mechanism of complement lectin-pathway activation revealing the role of serine protease MASP-1 as the exclusive activator of MASP-2. Proc. Natl. Acad. Sci. USA, 109, 10498-10503
Collaborations
Complement research
- Department of Biochemistry, Department of Chemistry, Eötvös University, Budapest
- 3rd Department of Internal Medicine, Semmelweis University , Budapest
- University of Aarhus, Denmark
- University Hospital, Bern, Switzerland
- University of British Columbia, Vancouver, Canada
Bioinformatics
- Department of Medical Chemistry, Molecular Biology and Pathobiochemistry , Semmelweis University, Budapest
- University of Michigan, USA
Instuments
- FPLC for protein purification
- QCM, DSC, FT IR
Teaching
- Undergraduate courses: Pázmány Péter Catholic University, Faculty of Information Technology and Bionics: biophysics, biochemistry and molecular biology
- Supervising B.Sc., M.Sc. and Ph.D. students at the Eötvös University and University of Technology
Leader
Péter Gál