Group leader: Katalin Monostory, PhD, DSc
The Human Organoid Laboratory forms part of the research group (principal investigator: Kornélia Szebényi, PhD).
Research interest:
Many undesired side-effects or therapeutic failure of drugs are the results of differences or alterations of drug metabolism. The team of the Metabolic Drug-interaction Research Group deals with interindividual differences in drug metabolism and elimination for more than 20 years. The research activities focus on the function and regulation of cytochrome P450 (CYP) enzymes, primarily involved in the metabolism of xenobiotics.
Biochemical, molecular biological and mass spectrometric approaches are applied for studying I) metabolism and pharmacokinetic interactions of drugs and drug-candidates under development, II) factors influencing the expression and function of CYP enzymes (hormonal status, disease, drug therapy, smoking), III) moreover, diagnostic approaches for patients’ drug metabolism capacity provide tools for personalized medication, IV and in vitro models are developed that can be used in toxicological and safety studies.
Main research projects:
1. Genetic and non-genetic factors influencing the activity of CYP enzymes
Pharmacogenetic analysis of CYP enzymes involved in drug metabolism contributes to personalized drug therapy. Phenotypes of drug-metabolizing enzymes are primarily influenced by genetic polymorphisms (loss-of-function or gain-of-function mutations, copy number variations) resulting in decreased or increased enzyme activity or complete loss of enzyme function. However, non-genetic factors (e.g. age, sex, enzyme inhibition and induction caused by drug interactions as well as diseases) can transiently decrease or increase metabolic activity leading to the phenotype different from that predicted from genotype. This phenoconversion can substantially modify the genotype-based estimation of the activities of drug-metabolizing enzymes.
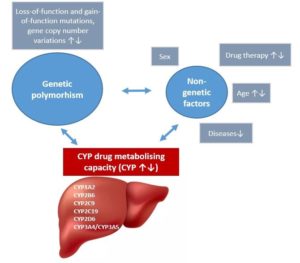
Impact of genetic and non-genetic factors on drug metabolism capacity.
The effects of clinically relevant polymorphic CYP allele-variants were investigated using liver tissues of human organ donors. Gene specific and accurate methods for genotyping and haplotype determination are crucial in estimation of the impact of polymorphic CYP alleles that required development of PCR-based methods for identification of CYP1A2, CYP2B6 and CYP2D6 variants. Besides genetic polymorphisms, several non-genetic factors including sex, drug therapies, chronic alcohol consumption were proved to significantly alter the phenotype predicted from CYP genotypes, for example in case of CYP1A2, CYP2B6, CYP2C9, CYP2C19, CYP2D6 enzymes (Fekete 2021 és 2022, Mangó 2022, Kiss 2018a és 2018b).
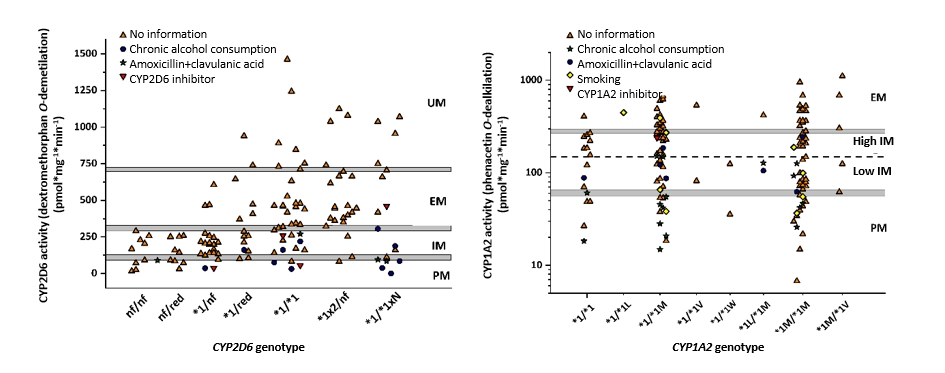
The effect of CYP genetic variability and non-genetic factors (CYP inhibition and induction; chronic alcohol consumption, amoxicillin+clavulanic acid therapy) on CYP2D6 and CYP1A2 enzyme activities. Due to the significant number of genotype variations, CYP2D6 activity-genotype relationships were evaluated based on PharmVar and CPIC recommendations. PM poor, IM intermediate, EM normal, UM ultra-rapid metabolizer phenotype; nf non-functional, red reduced functional allele, xN CYP gene multiplication
From the genotype based on clinically relevant CYP allelic variants, CYP enzyme activities can be inferred to various extent (0-67%). Non-genetic factors causing phenoconversion may be responsible for CYP genotype-phenotype differences (“mismatch”) (15-34%). In addition to CYP-selective inhibition and induction effects, we must be aware of non-CYP-specific factors, such as chronic alcohol consumption and amoxicillin+clavulanic acid treatment, which significantly reduced the expression and activity of several CYP enzymes.
Evaluation of CYP genotype based phenotype (CYP activity) estimation and of phenoconversion caused by non-genetic factors in human liver tissue samples.
| Identifying CYP phenotype (enzyme activity) influencing factors* | Genotype-phenotype „mismatch” | ||
| CYP genotype | Phenoconversion caused by non-genetic factors | ||
| CYP1A2 | ? | 25% | ? |
| CYP2B6 | 28% | 34% | 38% |
| CYP2C9 | 53% | 15% | 32% |
| CYP2C19 | 40% | 21% | 39% |
| CYP2D6 | 67% | 18% | 15% |
* frequency of hepatic CYP activities that can be explained by genetic or non-genetic factors
Modelling of cross-talk between sterol homeostasis and drug metabolism
The natural changes in hormonal status, or therapeutic steroids can cause substantial alteration in CYP gene expression and in CYP enzyme protein levels. The cross-talk between drug-metabolizing CYP enzymes and steroids such as dehydroepiandrosterone (DHEA), dexamethasone and cholesterol was investigated (Monostory 2005, Belic 2013, Kőhalmy 2007).
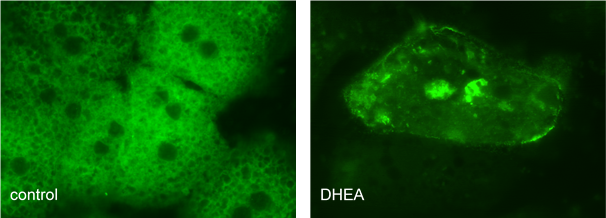
Dehydroepiandrosterone activates hepatic CAR (constitutive androstane receptor) nuclear receptor, which enters the nucleus and induces the transcriptions of CYP2B6, CYP2C9, CYP2C19 and CYP3A4 genes.
2. Strategy for personalized medication adjusted to patients’ drug-metabolizing capacity
CYPtestTM, the multi-step diagnostic system for the estimation of patients’ drug-metabolizing capacity, identifies polymorphic CYP alleles by DNA analysis (CYP-genotyping) and provides information about the current expression of key drug-metabolizing CYP enzymes by CYP-phenotyping (Temesvári 2012). CYPtestTM has been introduced for patients who are on multi-drug therapy or for those who particularly benefit from tailored medication by increasing drug efficacy and by significantly decreasing the risk of toxicity. The main focus is on the patients’ reduced or even extensive drug-metabolizing capacity (transplant recipients, psychiatric and neurologic patients as well as in patients suffering from liver dysfunction and cardiovascular diseases) which may potentially lead to therapeutic failure and severe adverse drug reactions. By recognizing poor or extensive drug metabolism, tailored medication adjusted to the patients’ drug-metabolizing capacity (by the optimization of the most appropriate drug and dosage) can minimize harmful side effects and ensure a more rational drug therapy and a more successful outcome.
Possibilities of personalized drug therapy
Systematic evaluation of the role of drug-metabolizing CYP enzymes in the metabolism of antipsychotics, mood-stabilizers and antiepileptics frequently applied in treatment of psychiatric and neurologic patients as well as of immunosuppressant drugs, the main components of transplant patients’ therapy, and drugs applied in anticancer therapy. The contribution of CYP polymorphisms (genetic and non-genetic variations) to inter-individual differences in the efficacy and toxicity of these drugs are estimated.
2.1. Oxidative metabolism of olanzapine, the widely used second-generation antipsychotic, is linked to CYP enzymes (mainly to the polymorphic CYP1A2 and to a lesser extent CYP2D6). The data regarding the impact of CYP1A2 polymorphisms on CYP1A2 activity are inconsistent in the literature, the CYP1A2 genetic variants are not appropriate for estimation of CYP1A2 enzyme activity; however, CYP1A2 mRNA expression in leukocytes appears to properly predict hepatic CYP1A2 activity. Drug metabolizing capacity based on CYP1A2 mRNA expression showed a close correlation with the blood level of olanzapine in the patients. Furthermore, smoking that can induce CYP1A2 transcription has been shown to lead to an increase in olanzapine metabolism in psychiatric patients.
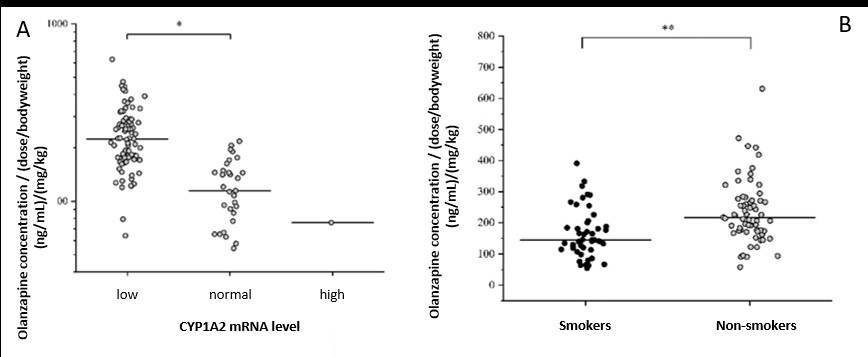
Effect of CYP1A2 mRNA expression on olanzapine plasma concentration in psychiatric patients (A) and the effect of patients’ smoking habits on olanzapine plasma concentrations (B). *P<0.001, **P<0.0001
The prediction model, based on the genetic and non-genetic factors affecting olanzapine blood concentration can be used in personalized dose adjustment, which can reduce the risk of the lack of the therapeutic effect or of the development of adverse side effects of olanzapine.
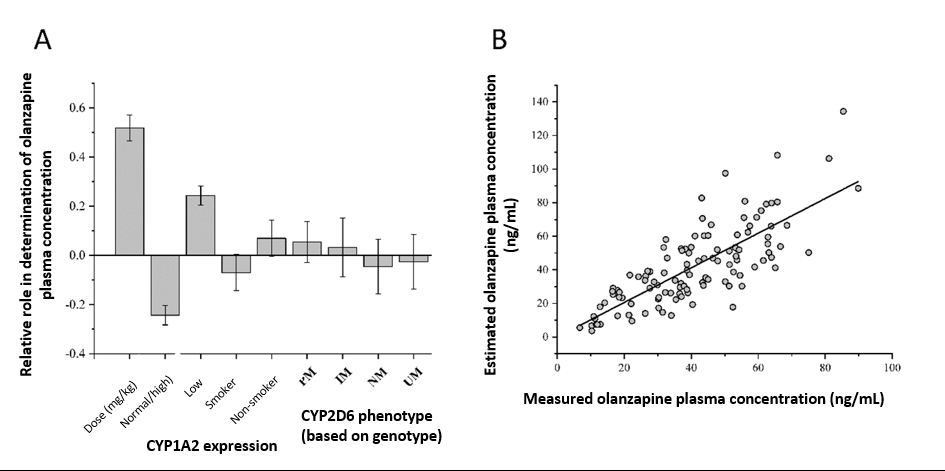
Factors influencing olanzapine plasma concentration. (A) Principal component analysis with input variables of olanzapine daily dose/patients’ bodyweight, patients’ CYP1A2 expression, smoking behaviour and CYP2D6 genotype-based phenotype; (B) Olanzapine plasma concentrations predicted by the partial least square (PLS) regression model. PM poor, IM intermediate, NM normal, UM ultrarapid metabolizers
2.2. The clinical consequences of decreased CYP2C9 function were investigated in children with epilepsy. It has been established that valproic acid, one of the first choices of antiepileptic drugs, is metabolized primarily by CYP2C9 in pediatric patients. Identification of loss-of-function mutations in CYP2C9 may lead to false prediction of a patient’s valproate metabolizing capacity, since CYP2C9 expression highly influences blood concentrations of valproate.
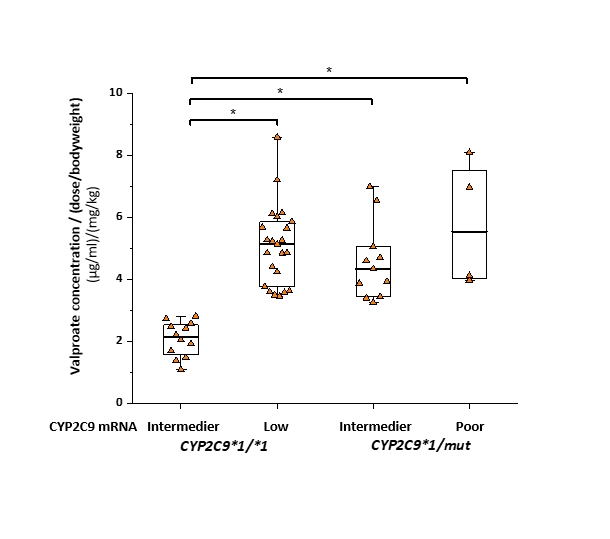
Serum concentrations of valproic acid in epileptic children with different CYP2C9-statuses. (CYP2C9*1/mut: heterozygous CYP2C9 genotype (CYP2C9*1/*2 or CYP2C9*1/*3); normal: intermediate CYP2C9 expressions; Low: low CYP2C9 expressions; *P <0.0001)
Patients’ CYP2C9-status guided dosing strategy for achieving the optimal blood concentration has been suggested (Tóth 2015, Monostory 2019).
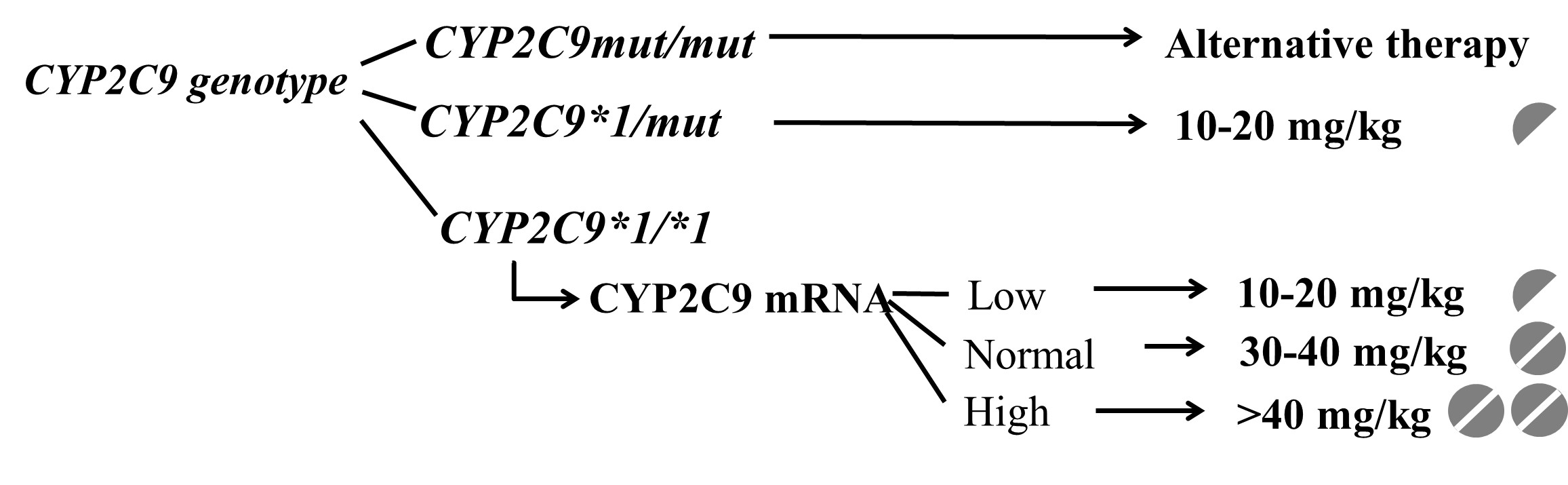
Valproic acid dose required for the therapeutic serum concentrations in epileptic children with various CYP2C9-statuses.
CYP2C9-guided (CYP2C9 genotype and CYP2C9 expression) treatment significantly reduced the ratio of patients out of the range of target valproate blood concentrations, the ratio of patients with abnormal serum alkaline phosphatase levels and the incidence of serious side effects, notably hyperammonemia (Bűdi 2015, Monostory 2019).
2.3. The incidence of adverse reactions in the anticonvulsant clonazepam therapy is highly attributed to the inter-individual variability in clonazepam metabolism by CYP3A and NAT2 (N-acetyl transferase 2) enzymes. The patients’ CYP3A4 expression was found to be the major determinant of clonazepam plasma concentrations; whereas CYP3A5 genotype and NAT2 acetylator phenotype did not influence the steady state levels of clonazepam. (Tóth 2016).
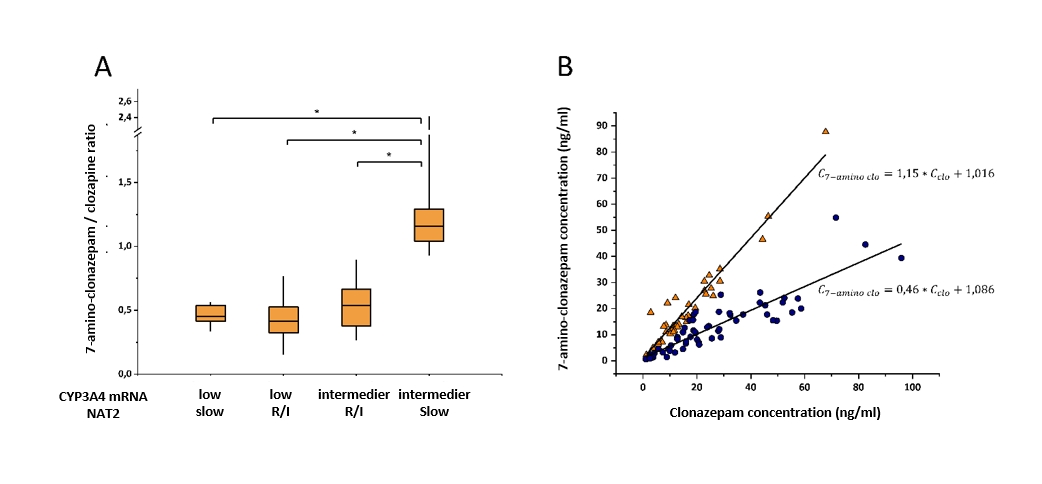
Effect of CYP3A4 expression and NAT2 acetylator phenotype on 7-aminoclonazepam blood concentration in psychiatric patients. A) 7-aminoclonazepam/clonazepam blood level ratio in patients with different CYP3A4 expression and NAT2 acetylator phenotypes; B) linear model of 7-aminoclonazepam – clonazepam blood levels in slow acetylators with CYP3A4 intermediate expression (triangle) and all other CYP3A4-NAT2 patients (circle). R/I rapid/intermediate acetilation phenotype; Cclo clonazepam plasma concentration, C7-amino clo 7-amino-clonazepam plasma concentration; *P<0.0001
However, normal CYP3A4 expression and slow NAT2 acetylation phenotype evoking high plasma concentration ratio of 7-amino-clonazepam and clonazepam, may account for low efficacy or withdrawal symptoms of clonazepam. Prospective assaying of CYP3A4 expression and NAT2 acetylation phenotype can better identify the patients with high risk of adverse reactions and can facilitate the improvement of personalized clonazepam therapy and withdrawal regimen.
2.4. The atypical antipsychotic clozapine is effective in treatment-resistant schizophrenia; however, the success or failure of clozapine therapy is substantially affected by the variables that impact clozapine blood concentration. CYP3A4 expression was found to be the major determinant of normalized clozapine concentration, particularly in patients expressing CYP1A2 at relatively low level. The functional CYP3A5*1 allele seemed to influence clozapine concentrations in those patients who expressed CYP3A4 at low levels. Strong association was observed between the metabolite/clozapine ratios and CYP3A4 mRNA levels, which confirmed the primary role of CYP3A4 in clozapine metabolism (Tóth 2017).
2.5. CYP2D6, which is the dominant enzyme in metabolism of the antipsychotic aripiprazole and in dehydroaripiprazole , was inhibited by concomitant treatment with the antipsychotic risperidone, and with metoprolol or propranolol applied for mitigation of the side effect (akathisia), and caused an increase in aripiprazole blood levels in patients carrying at least one wild-type CYP2D6*1 allele. Risperidone and its 9-hydroxy metabolite inhibited the formation of both dehydroaripiprazole and hydroxyaripiprazole, which means that metabolism of risperidone did not eliminate its CYP2D6 inhibitory effect. Metoprolol and propranolol only blocked the formation of the pharmacologically active dehydroaripiprazole and thus directed the metabolism of aripiprazole towards the pathways resulting in inactivation (Kiss 2020).
2.6. CYP3A-status, taking both CYP3A4 expression and CYP3A5 genotype into account, influences recipients’ calcineurin inhibitor therapy after transplantation. In liver transplant patients, CYP3A-status of the donor liver contributes to the recipient’s blood concentrations of ciclosporin and tacrolimus. It has been reported that patients transplanted with liver grafts from low or high expressers or with grafts carrying functional CYP3A5*1 allele required substantial modification of the initial calcineurin inhibitor dose. Donor livers’ CYP3A-status can better identify the risk of calcineurin inhibitor over- or underexposure, and may contribute to the avoidance of misdosing-induced graft injury in the early postoperative period (Monostory 2015).
The time for achieving therapeutic tacrolimus concentration was significantly reduced, confirming potential benefit of initial tacrolimus therapy adjusted to donor’s CYP3A-status over classical clinical practice of tacrolimus concentration guided treatment (4 vs 8 days, P<0.0001). Acute rejection episodes (3.6% vs 23.8%, P<0.0001) and tacrolimus induced nephrotoxicity (8% vs 27%, P=0.0004) were less frequent in patients on CYP3A-status guided tacrolimus therapy (Csikány 2021).
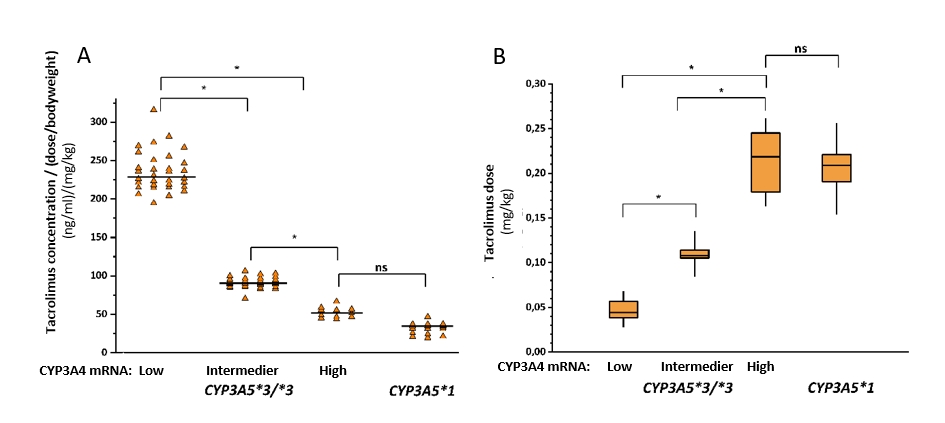
A) Influence of the donors’ CYP3A-status (CYP3A5 genotypes and CYP3A4 expression) on the blood concentrations of tacrolimus in liver transplant patients. B) Dose requirements of recipients in the course of CYP3A-status of the liver donors are presented. (Low, Normal, High: the levels of CYP3A4 expression; ns: not statistically significant); *P<0,0001
Previous experience in liver transplant patients contributed to personalized immunosuppressive therapy in heart transplant patients. In the early postoperative immunosuppressive therapy, high-dose corticosteroid treatment affected drug metabolizing capacity of patients by regulating transcription of CYP3A enzymes. In the 15-month post-operative period, the research group investigated the alterations in heart transplant recipient’s drug metabolizing capacity and phenoconverting factors that modified pharmacokinetics of immunosuppressive agents (primarily tacrolimus) (Déri 2021).
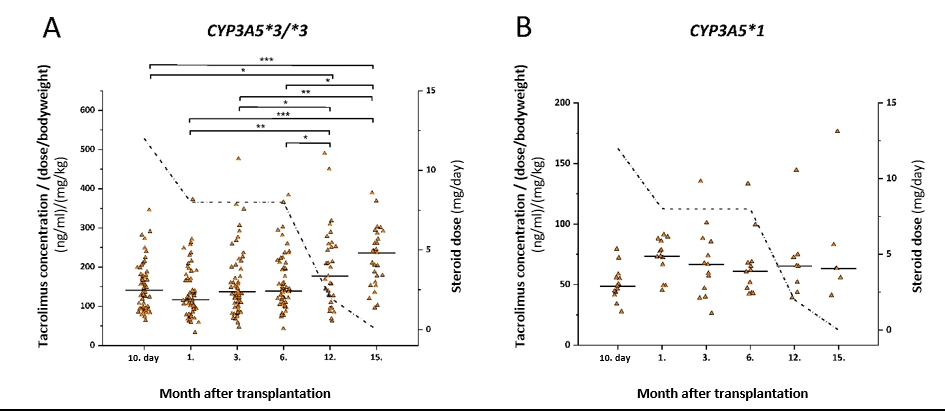
The dose-corrected tacrolimus blood concentrations in heart transplant recipients with CYP3A5*3/*3 genotype (A) and recipients carrying CYP3A5*1 allele (B) in a 15-month period after transplantation. The dotted line represents the daily dose of corticosteroid. *P<0.05, **P<0.001, ***P<0.0001
2.7. Among the substrates of CYP2B6, the antitumor cyclophosphamide alkylating agent must be highlighted, which is used in cancer therapy, including neuroblastoma affecting infants and young children. The CYP2B6 metabolizing capacity of neuroblastoma patients was estimated on the basis of CYP2B6 genotype and adverse effects following cyclophosphamide therapy were evaluated. A correlation was found between CYP2B6 metabolizing capacity of neuroblastoma patients treated with cyclophosphamide and severe hematotoxicity as well as liver injury developing after cyclophosphamide therapy. Patient’s CYP2B6 genotype can predict development of side effects following cyclophosphamide treatment and adjuvant drug therapy can be started in time to alleviate toxic events (Mangó 2023).
3. Development of in vitro models
Various in vitro models have been developed for in vitro testing of drugs that are useful in pharmacological, pharmacokinetic, and toxicity studies.
3.1. Human induced pluripotent stem cell-derived brain organoids in pharmacology and toxicology studies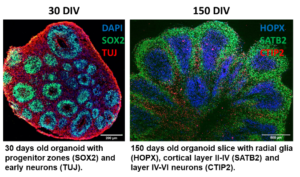
Aging societies face new challenges, one of the most serious being the increasing incidence of cancer and neurodegenerative diseases. In order to successfully combat these diseases, researchers are working to map the human body as accurately as possible when developing model systems. In the case of the brain, cerebral organoids are able to do this, properly reproducing not only the cell types that make up the cortical plate, but also their structural arrangement. From these organoids, slice cultures are created that can be sustained for up to one year when grown at the air-liquid interface. Thus, mature neuronal and glia cell types can develop that allow the study of specific pathological changes in neurodegenerative diseases such as amyotrophic lateral sclerosis with frontotemporal dementia (ALS / FTD) and screening of drug molecules that can potentialy prevent or slow down the disease (Szebényi 2021).
3.2. Pharmacokinetic variations leading to therapy resistence in cancer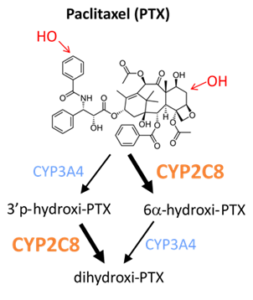
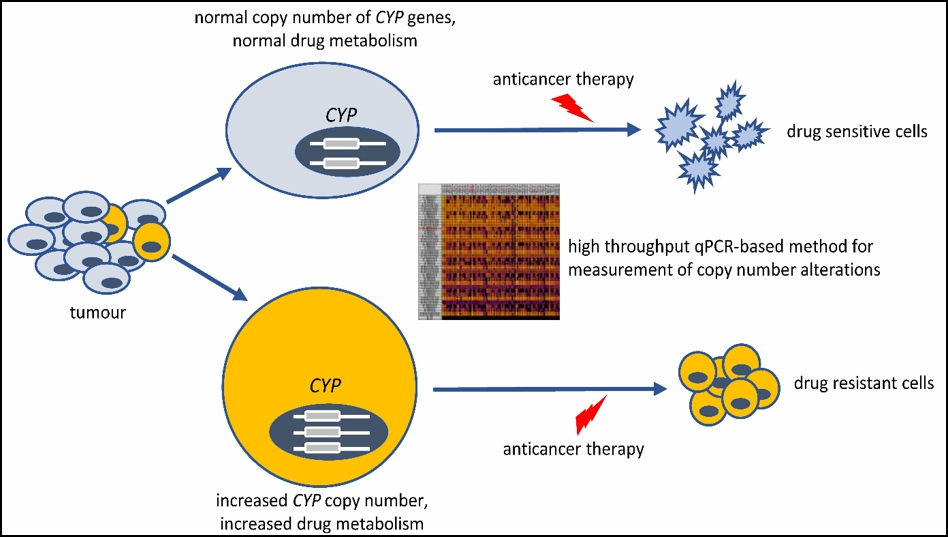
The research group in collaboration with National Korányi Institute of Pulmonology and Drug Resistance Research Group investigates the role of pharmacokinetic variability in drug resistance that highly impact clinical outcome of cancer therapy. Resistance to antitumor therapies is a major obstacle to effective treatment and is largely responsible for the high mortality rate. Due to the genetic polymorphisms of drug-metabolizing CYP enzymes, some resistance mechanisms are related to the pharmacokinetic variability of the metabolism of anticancer agents. Alterations in CYP copy numbers in tumor cells are thought to contribute to the survival of resistant cells. A high-throughput quantitative polymerase chain reaction (qPCR)-based method was developed for the detection of CYP copy number changes in tumors (Incze 2023). In addition, they created resistant cell models in which they can investigate the influence of CYP2C8 and CYP3A4 overexpression (paclitaxel metabolizing enzymes) on paclitaxel cytotoxicity. The high-throughput qPCR-based method can become an alternative approach to next-generation sequencing in routine clinical practice, and the identification of altered CYP copy numbers can be a promising biomarker in the case of therapy-resistant tumors.
3.3. Development of biomarkers for toxicological and safety studies
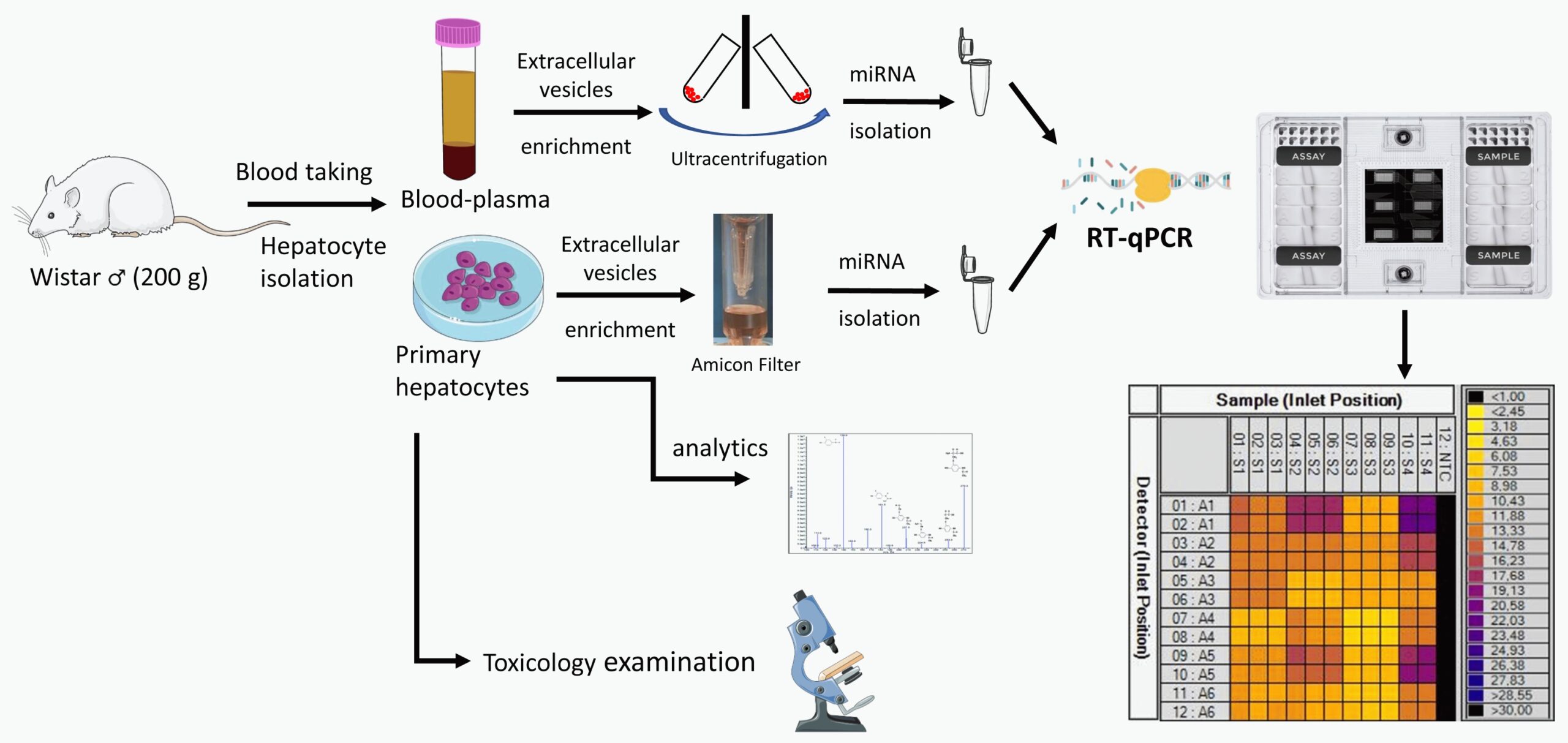
The goal of the project is to identify extracellular microRNA (miRNA) based biomarkers that can indicate organ-specific injury earlier than classical toxicological markers. The research and development aims early detection of liver, kidney or cardiovascular injury within the framework of classic toxicological tests. The continuous monitoring of extracellular miRNA-based biomarkers, which can be incorporated into single-dose but especially repeated-dose studies, creates an opportunity to indicate organ-specific toxic effect even before the classical toxicological endpoints appear. miRNAs are RNA molecules that do not code proteins, but can bind to the 3′ untranslated region of mRNAs and inhibit its transcription and induce its degradation. A functionally determined proportion of miRNAs are found in extracellular vesicles. Extracellular vesicles are found in all body fluids and their use as non-invasive or minimally invasive biomarkers has great potential. Analytical detection of miRNA-based potential biomarkers is developed using in vitro cell culture models (e.g. primary hepatocyte cells) and toxicological tests with model compounds (e.g. acetaminophen). Validation of the new biomarker candidates is planned for miRNA-based detection of induced liver and kidney damage and cardiotoxicity in blood plasma and urine in in vivo animal models and their comparison with classical toxicological endpoints (biochemical markers, organ weight, histopathology).
3.4. In vitro model for testing immunosuppression efficacy
Ex vivo immunostimulation model has been developed using peripheral blood mononuclear cells to characterize immunosuppressive agents on the basis of their inhibitory effects on the expression of pro-inflammatory cytokines. The model is to be used to assaying the efficacy of immunosuppressive therapy after organ transplantation and the immunological background of acute rejection events.
Lab Equipments:
- Fluorescent microscope
- Cell-culture laboratory equipped with CO2 incubator, Esco Class II Biological Safety Cabinet, microscope
- HPLC UV-VIS, radiodetector
- Fluorimeter
- NanoDrop 1000 Spectrophotometer
- Real-time PCR and end-point PCR systems (BioRad)
- Western blot systems
- Fluidigm BiomarkTM HD high throughput PCR system
- Bio-Rad Droplet Digital PCR systems
- QuantStudio 5 real-time PCR systems
- Agilent 4200 TapeStation nucleic acid analyzer
- Ultracentrifuge
Collaborations:
International
- Faculty of Medicine, University of Ljubljana, Ljubljana, Slovenia – The cross-talk between cholesterol homeostasis and drug- metabolism
- Lek Pharmaceuticals d.d., Menges. Slovenia – Modelling of relationship between pharmacokinetic behavior of medicines and variability of CYP enzymes
- Palacky University, Olomouc, Czech Republic – The effect of the steroid type compounds (dexamethasone, dehydroepiandrosterone) on drug-metabolizing cytochrome P450 enzymes
- INSERM (Institut National de la Santé et de la Recherche Médicale) U632, Montpellier, France – The role of nuclear receptors in the regulation of CYP enzymes
- University of Cambridge, Department of Clinical Neurosciences, Lakatos Lab – Development of ALS/FTD organoid models
- Water Research Institute, National Research Council, Rome, Italy – Drug levels in the Danube near Budapest and its area
National
- Genome Stability Research Group, HUN-REN, RCNS, Institute of Molecular Life Sciences, Budapest
- Drug Resistance Research Group, HUN-REN, RCNS, Institute of Molecular Life Sciences, Budapest
- Biological Nanochemistry Research Group, HUN-REN, RCNS, Institute of Materials and Environmental Chemistry, Budapest
- Biomolecular Self-assembly Research Group, HUN-REN, RCNS, Institute of Materials and Environmental Chemistry, Budapest
- Biomolecular Interactions Research Group, HUN-REN, RCNS, Institute of Organic Chemistry, Budapest
- Centre for Structural Sciences, HUN-REN, RCNS, Budapest
- Department of Surgery ,Transplantation and Gastroenterology, Semmelweis University, Budapest – Drug metabolism in transplant patients; Prevention of toxicity resulted from ciclosporin metabolism
- Heart and Vascular Center, Semmelweis University, Budapest – Drug metabolism in transplant patients; Prevention of toxicity resulted from immunosuppressant metabolism
- Department of Psychiatry and Psychotherapy, Semmelweis University, – Possibilities of personalized antipsychotic therapy
- Department of Pediatrics, Semmelweis University – CYP copy number variations in tumorous tissues
- Madarász Street Department, Heim Pál Children’s Hospital, Budapest – Development of personalized antiepileptic therapy
- Department of Food Hygiene, University of Veterinary Medicine, Budapest – Pharmacokinetic study of antiviral agents
- Department of Transplantation, University of Debrecen, Debrecen – Investigation of the drug-metabolizing capacity of kidney transplant patients
- Department of Internal Medicine, University of Szeged, Szeged – Development of personalized therapy for patients with inflammatory bowel diseases
- St László Hospital, Budapest – Personalized therapy of bone-marrow transplant patients
- National Korányi Institute of Pulmonology – The mechanisms of therapy resistance, designing personalized therapies
- Centre for Energy Research, Institute of Technical Physics and Materials Science, Microsystems Laboratory – Establishing physiological organoid culture technology
- “Toxi-Coop” Toxicological Research Center Zrt., Budapest
Education/training:
- Hungarian University of Agriculture and Life Sciences, Budapest
- Semmelweis University, Budapest
- Eötvös Loránd University, Budapest
- Budapest University of Technology and Economics, Budapest
PhD students in the last 10 years:
Nóra Csikány (2022)
Máté Tamás Déri (2022)
Ádám Ferenc Kiss (2020)
Katalin Tóth (2018)
Pálma Porrogi (2016)
Manna Temesvári (2013)
Selected publications:
Incze E, Mangó K, Fekete F, Kiss Á. F, Póti Á, Harkó T, Moldvay J, Szüts D & Monostory, K: Potential association of cytochrome P450 copy number alteration in tumour with chemotherapy resistance in lung adenocarcinoma patients. International Journal of Molecular Sciences, 24(17), 13380, 2023
Fekete F, Menus Á, Tóth K, Kiss Á. F, Minus A, Sirok D, Belič A, Póti Á, Csukly G & Monostory, K: CYP1A2 expression rather than genotype is associated with olanzapine concentration in psychiatric patients. Scientific Reports, 13(1), 18507, 2023
Mangó K, Fekete F, Kiss Á. F, Erdős R, Fekete J. T, Bűdi T, Bruckner E, Garami M, Micsik T & Monostory, K: Association between CYP2B6 genetic variability and cyclophosphamide therapy in pediatric patients with neuroblastoma. Scientific Reports, 13(1), 11770, 2023
Mangó K, Kiss Á. F, Fekete F, Erdős R, & Monostory K: CYP2B6 allelic variants and non-genetic factors influence CYP2B6 enzyme function. Scientific Reports, 12(1), 2984, 2022
Fekete F, Mangó K, Minus A, Tóth K, & Monostory K: CYP1A2 mRNA Expression Rather than Genetic Variants Indicate Hepatic CYP1A2 Activity. Pharmaceutics, 14(3), 532, 2022
Szebényi K, Wenger LMD, Sun Y, Dunn AWE, Limegrover CA, Gibbons GM, Conci E, Paulsen O, Mierau SB, Balmus G and Lakatos A: Human ALS/FTD brain organoid slice cultures display distinct early astrocyte and targetable neuronal pathology. Nature Neuroscience 24: 1542–1554, 2021
Déri M, Szakál-Tóth Zs, Fekete F, Mangó K, Incze E, Minus A, Merkely B, Sax B, Monostory K: CYP3A-status is associated with blood concentration and dose-requirement of tacrolimus in heart transplant recipients. Scientific Reports 11: 21389, 2021
Fekete F, Mangó K, Déri M, Incze E, Minus A, Monostory K: Impact of genetic and non-genetic factors on hepatic CYP2C9 expression and activity in Hungarian subjects. Scientific Reports 11: 17081, 2021
Csikány N, Kiss Á, Déri M, Fekete F, Minus A, Tóth K, Temesvári M, Sárváry E, Bihari L, Gerlei Zs, Kóbori L, Monostory K: Clinical significance of personalized tacrolimus dosing by adjusting to donor CYP3A-status in liver transplant recipients. British Journal of Clinical Pharmacology 87: 1790–1800, 2021
Menus Á, Kiss Á, Tóth K, Sirok D, Déri M, Fekete F, Csukly G, és Monostory K: Association of clozapine-related metabolic disturbances with CYP3A4 expression in patients with schizophrenia. Scientific Reports 10: 212383, 2020
Déri M. T, Kiss Á. F, Tóth K, Paulik J, Sárváry E, Kóbori L, Monostory K: End-stage renal disease reduces the expression of drug-metabolizing cytochrome P450s. Pharmacological Reports 72: 1695–1705, 2020
Kiss Á, Menus Á, Tóth K, Déri M, Sirok D, Gabri E, Belic A, Csukly G, Bitter I, Monostory K: Phenoconversion of CYP2D6 by inhibitors modifies aripiprazole exposure. European Archives of Psychiatry and Clinical Neuroscience 270: 71–82, 2019
Monostory K, Nagy A, Tóth K, Bűdi T, Kiss Á, Déri M, Csukly G: Relevance of CYP2C9 function in valproate therapy. Current Neuropharmacology 17: 99–106, 2019
Kiss Á, Tóth K, Juhász C, Temesvári M, Paulik J, Hirka G, Monostory K: Is CYP2D6 phenotype predictable from CYP2D6 genotype? Microchemical Journal 136: 209-214, 2018a
Kiss ÁF, Vaskó D, Déri MT, Tóth K, Monostory K: Combination of CYP2C19 genotype with non-genetic factors evoking phenoconversion improves phenotype prediction. Pharmacological Reports 70: 525–532, 2018b
Tóth K, Csukly G, Sirok D, Belic A, Kiss Á, Háfra E, Déri M, Menus Á, Bitter I, Monostory K: Potential role of patients’ CYP3A-status in clozapine pharmacokinetics. International Journal of Neuropsychopharmacology 20: 529-537, 2017
Tóth K, Csukly G, Sirok D, Belic A, Háfra E, Kiss Á, Déri M, Menus Á, Bitter I, Monostory K: Optimization of clonazepam therapy adjusted to patient’s CYP3A-status and NAT2 genotype. International Journal of Neuropsychopharmacology 19: 1-9, 2016
Bűdi T, Tóth K, Nagy A, Szever Z, Kiss Á, Temesvári M, Háfra E, Garami M, Tapodi A, Monostory K: Clinical significance of CYP2C9-status guided valproic acid therapy in children. Epilepsia 56: 849-855, 2015
Monostory K, Tóth K, Kiss Á, Háfra E, Csikány N, Paulik J, Sárváry E, Kóbori L: Personalizing calcineurin inhibitor therapy by adjusting to donor CYP3A-status in liver transplant patients
British Journal of Clinical Pharmacology 80: 1429-1437, 2015
Tóth K, Bűdi T, Kiss Á, Temesvári M, Háfra E, Nagy A, Szever Z, Monostory K: Phenoconversion of CYP2C9 in epilepsy limits the predictive value of CYP2C9 genotype in optimizing valproate therapy. Personalized Medicine 12: 199-207, 2015
Szebényi K, Péntek A, Erdei Z, Várady G, Orbán TI, Sarkadi B, Apáti Á: Efficient generation of human embryonic stem cell-derived cardiac progenitors based on tissue-specific enhanced green fluorescence protein expression. Tissue Engineering, Part C: Methods 21: 35-45, 2015
Szebényi K, Füredi A, Kolacsek O, Csohány R, Prókai Á, Kis-Petik K, Szabó A, Bősze Z, Bender B, Tóvári J, Enyedi Á, Orbán TI, Apáti Á, Sarkadi B: Visualization of Calcium Dynamics in Kidney Proximal Tubules. Journal of the American Society of Nephrology 26: 2731-2740, 2015
Szebényi K, Füredi A, Kolacsek O, Pergel E, Bősze Z, Bender B, Vajdovich P, Tóvári J, Homolya L, Szakács G, Héja L, Enyedi Á, Sarkadi B, Apáti Á, Orbán TI: Generation of a Homozygous Transgenic Rat Strain Stably Expressing a Calcium Sensor Protein for Direct Examination of Calcium Signaling. Scientific Reports 5: 12645, 2015
Belic A, Tóth K, Vrzal R, Temesvári M, Porrogi P, Orbán E, Rozman D, Dvorak Z, Monostory K: Dehydroepiandrosterone post-transcriptionally modifies CYP1A2 induction involving androgen receptor. Chemico-Biological Interactions 203: 597-603, 2013
Temesvári M, Kóbori L, Paulik J, Sárváry E, Belic A, Monostory K: Estimation of drug-metabolizing capacity by cytochrome P450 genotyping and expression. Journal of Pharmacology and Experimental Therapeutics 341: 294-305, 2012
Temesvári M, Paulik J, Kóbori L, Monostory K: High-resolution melting curve analysis to establish CYP2C19*2 single nucleotide polymorphism: comparison with hydrolysis SNP analysis. Molecular and Cellular Probes 25: 130-133, 2011
Monostory K, Dvorak Z: Steroid regulation of drug-metabolizing cytochromes P450. Current Drug Metabolism 12: 154-172, 2011
Rezen T, Rozman D, Pascussi J-M, Monostory K: Interplay between cholesterol and drug metabolism. Biochim Biophys Acta – Proteins and Proteomics 1814: 146-160, 2011
Rozman D, Monostory K: Perspectives of the non-statin hypolipidemic agents. Pharmacology and Therapeutics 127: 19-40, 2010
Belic A, Temesvári M, Kőhalmy K, Vrzal R, Dvorak Z, Rozman D, Monostory K: Investigation of the CYP2C9 induction profile in human hepatocytes by combining experimental and modelling approaches. Current Drug Metabolism 10: 457-461, 2009
Monostory K, Pascussi J-M, Kóbori L, Dvorak Z: Hormonal regulation of CYP1A expression. Drug Metabolism Reviews 41: 547-572, 2009
Monostory K, Pascussi J-M, Szabó P, Temesvári M, Kőhalmy K, Acimovic J, Kocjan D, Kuzman D, Wilzewski B, Bernhardt R, Kóbori L, Rozman D: Drug-interaction potential of 2-((3,4-(dichlorophenethyl(propyl)amino)-1-(pyridin-3-yl)ethanol (LK-935), the novel non-statin type cholesterol lowering agent. Drug Metabolism and Disposition 37: 375-385, 2009
Kóbori L, Kőhalmy K, Porrogi P, Sárváry E, Gerlei Zs, Fazakas J, Nagy P, Járay J, Monostory K: Drug-induced liver graft toxicity caused by cytochrome P450 poor metabolism. British Journal of Clinical Pharmacology 65: 428-436, 2008
Kőhalmy K, Tamási V, Kóbori L, Sárváry E, Pascussi J-M, Porrogi P, Rozman D, Prough RA, Meyer UA, Monostory K: Dehydroepiandrosterone induces human CYP2B6 through the constitutive androstane receptor. Drug Metabolism and Disposition 35: 1495-1501, 2007
Monostory K, Kőhalmy K, Prough, RA, Kóbori L, Vereczkey L: The effect of synthetic glucocorticoid, dexamethasone on CYP1A1 inducibility in adult rat and human hepatocytes. FEBS Letters 579: 229-235, 2005
Monostory K, Hazai E, Vereczkey L: Inhibition of cytochrome P450 enzymes participating in p-nitrophenol hydroxylation by drugs known as CYP2E1 inhibitors. Chemico-Biological Interactions 147: 331-340, 2004
Szűcs G, Tamási V, Laczay P, Monostory K: Biochemical background of toxic interaction between tiamulin and monensin. Chemico-Biological Interactions 147: 151-161, 2004
Tamási V, Hazai E, Porsmyr-Palmertz M, Ingelman-Sundberg M, Vereczkey L, Monostory K: GYKI-47261, a new AMPA antagonist is a CYP2E1 inducer. Drug Metabolism and Disposition 31:1310-1314, 2003
Tamási V, Vereczkey L, Falus A, Monostory K: Some aspects of interindividual variations in the metabolism of xenobiotics. Inflammation Research 52:322-333, 2003
Hazai E, Vereczkey L, Monostory K: Reduction of toxic metabolite formation of acetaminophen. Biochemical Biophysical Research Communications 291: 1089-1094, 2002
Tamási V, Kiss Á, Dobozy O, Falus A, Vereczkey L, Monostory K: The effect of dexamethasone on P450 activities in regenerating liver. Biochemical Biophysical Research Communications 286: 239-242, 2001
Monostory K, Vereczkey L, Lévai F, Szatmári I: Iprifalvone as an inhibitor of human cytochrome P450 enzymes. British Journal of Pharmacology 123: 605-610, 1998
Monostory K, Jemnitz K, Vereczkey L, Czira G: Species differences in metabolism of panomifene, an analogue to tamoxifen. Drug Metabolism and Disposition 25: 1370-1378, 1997
Monostory K, Vereczkey L: The effect of phenobarbital and dexamethasone coadministration on the activity of rat liver P450 system. Biochemical Biophysical Research Commununications 203: 351-358, 1994









Leader
Katalin Monostory