The complement system is a powerful effector branch of the innate immunity in the blood and in other body fluids. The complement system, like the blood coagulation, is a proteolytic cascade system which can recognize, label and eliminate invading pathogens (e.g. bacteria, viruses, fungi) and dangerously altered host cells (e.g apoptotic cells, virus infected cells, cancer cells). An intact complement system is necessary for maintaining the homeostasis of the body, however, uncontrolled overactivation of the system leads to the development of sever disease conditions. The inappropriate complement activation is involved in myocardial infarction, stroke, age-related macular degeneration and Alzheimer-disease. It is of utmost importance to reveal the exact mechanism of complement activation and identify the proper drug targets for treatment of complement-related diseases.
The complement system can be activated by three different routes: the classical, the lectin and the alternative pathway. The classical and the lectin pathways are initiated by pattern recognition molecules and associated serine proteases. The third pathway, the alternative pathway, although it can initiate on its own, provides a powerful amplification loop for the classical and the lectin pathways. When a pathogen enters into the bloodstream the alternative pathway is responsible for the 80-90% of the net complement activation on the surface of the microorganism.
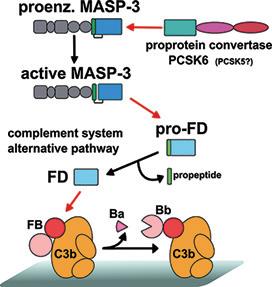
The alternative pathway was discovered in the 1950s, but important questions have remained unanswered until recently. The components of the complement system are synthesized as zymogens, and limited proteolysis is necessary for their activation. Factor D (FD), the first protease of the alternative pathway, is an exception, because it circulates as a cleaved component even before the appearance of any danger signal. Recently, researchers at the Institute of Enzymology RCNS (TTK) led by Péter Gál have discovered that FD is cleaved by MASP-3 (mannose-binding lectin-associated serine protease 3) in the circulation. It turned out, however, that MASP-3 is also present in its cleaved activated form in the bloodstream, like FD. The question aroused: What is the physiological activator of MASP-3? Researchers at RCNS discovered a hitherto hidden connection between two proteolytic systems in the body: the proprotein convertases and the complement system. Proprotein convertases are vital enzymes responsible for processing of multiple precursor proteins such as hormones, receptors, adhesion molecules, etc. The researchers showed that MASP-3 is activated by PACE4 (PCSK6), a proprotein convertase present in the circulation. In other words, the researchers discovered a new component of the complement system, the top activator of the alternative pathway, which is a proprotein convertase.
This surprising discovery highlights that the proteolytic systems are interconnected in the body even to a greater extent than it was previously thought. Proprotein convertases are new enzymes in respect of innate immunity. It was already known that there are cross-talks between the blood coagulation, fibrinolytic and complement systems. The finding that a proprotein convertase is involved in the complement activation opens up new perspectives in studying the mechanism of complement activation and also in identifying new drug targets for treating complement-related diseases. The discovery of the Hungarian scientists was published in The Journal of Immunology (the official journal of The American Association of Immunologists), as a “Top read” in the May issue (J Immunol. 2021 May 1;206(9):2198-2205).